The geometric (H/D) isotope effect in porphycene: grid-based Born–Oppenheimer vibrational wavefunctions vs. multi-component molecular orbital theory
Abstract
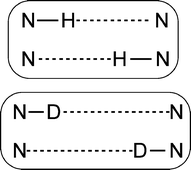
Two approaches for the determination of the primary and secondary geometric isotope effect are compared for the exemplary porphyrinoid system porphycene, which has two intramolecular hydrogen bonds. A three-dimensional Born–Oppenheimer potential energy surface is calculated in terms of the symmetric and antisymmetric N–H stretching as well as a low-frequency hydrogen bond vibrational normal mode coordinate. From the respective ground-state nuclear wavefunction the quantum correction to the classical equilibrium geometry is determined. Further, geometry optimization within a full-dimensional multi-component molecular orbital (MC_MO) type calculation, which treats both the electrons and the hydrogen-bonded

 Please wait while we load your content...
Please wait while we load your content...