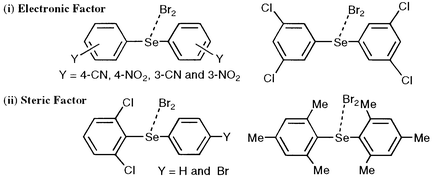
Diaryl selenides that yield molecular complexes (MC) with bromine are prepared by modulating the effective electronegativity of the selenium atom and the steric environment around the atom in diphenyl selenide (1) with various substituents at the 2-, 3- and/or 4-positions. Halogen induced 1H and 13C NMR chemical shifts of the diaryl selenides are examined. The chlorine and iodine adducts of the selenides are shown to be trigonal bipyramidal adducts (TB) and MC, respectively. In the case of bromine adducts, the structures of (3- and 4-YC6H4)2SeBr2 are demonstrated to be MC if Y is CN and NO2, contrary to the general rule: they are TB if Y is less electron-withdrawing than the ethoxycarbonyl group. The CN and NO2 groups increase the effective electronegativity of the Se atom in (3- and 4-YC6H4)2Se and do not give
TB with bromine. The four chloro groups at all meta-positions in 1 are also effective for MC formation. However, 3,5-(O2N)2C6H3SeBr2Ph is TB. Ab initio MO calculations show that the structures of (3-O2NC6H4)2Se and 3,5-(O2N)2C6H3SePh are close to the C2 and Cs symmetries, respectively, which reveals that the conformational change is also important when the structures of the bromine adducts are determined. The steric congestion must be more severe for TB formation than for MC formation. The structures of 2,6-Cl2C6H3SeBr2C6H4Y-p (Y = H and Br) are MC, which shows that the steric effect of 2,6-Cl2C6H3 group is effective for MC formation. (2-MeC6H4)2SeBr2
and (2-ClC6H4)2SeBr2 are TB and MC, respectively: the electronic effect of the Cl group must play an additional role in the MC formation since the bulkiness of the Me and Cl groups are expected to be similar. The bromine adduct of (2,4,6-Me3C6H2)2Se is also concluded to be MC: the steric effect of the four Me groups at the ortho-positions is large enough to give MC with bromine. The oxidation potentials (Eox) of the diaryl selenides explain well the structures of the bromine adducts: the electronic effect is directly correlated with Eox and the importance of the steric effect has been brought into sharp relief by Eox. Results of MO calculations support the outline of the observations. The structural behavior of some halogen adducts such as 2,6-Cl2C6H3SeCl2C6H4Y-p
(TB: Y = H and Br) is also discussed in some detail.

 Please wait while we load your content...
Please wait while we load your content...