Characterization of organic polyselenide ions in N,N-dimethylacetamide
Abstract
The
reactivity of selenolate ions RSe− [R = Ph (1), PhCH2 (2)] towards solid selenium has been investigated by UV-visible spectrophotometry in N,N-dimethylacetamide. After the slow formation of RSe2−, further addition of Se yielded
RSe3− and RSe4−
species, which partly disproportionated (RSe3−, 30% RSe4−, 50%) into diselanes RSe2R (a) and polyselenide
ions Se42− and Se62−, respectively. These equilibria were readily attained by the reactions between electrogenerated
Sex2−
ions (x = 4, 6) and substrates 1a and 2a. Visible spectra of RSe3−
and RSe4− ions (380 < λ < 580 nm)
were calculated [λmax = 420 (1), 400 nm (2)]. The formation of RSe2R as major products in the RSezR mixtures
obtained by the nucleophilic reactions Se42− + PhCH2Br (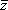 = 2.17) or CH3I (
= 2.17) or CH3I (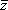 = 2.48) on a preparative scale agrees
with the
disproportionation of intermediate RSey− ions.
= 2.48) on a preparative scale agrees
with the
disproportionation of intermediate RSey− ions.

 Please wait while we load your content...
Please wait while we load your content...