Abstract
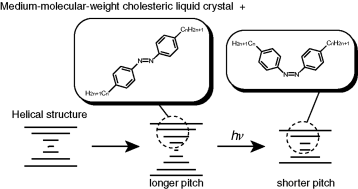
The effects of small amounts of achiral photochromic dopants, 4,4′-di-n-alkylazobenzenes, with various lengths of substituted alkyl chain (CnH2n + 1 ∶ n = 4, 7, 11, 12, 13, 14, 15, 16) on the helical pitch of a cholesteric liquid crystal (CLC) with ca. 1000 molecular weight were investigated in order to utilise the cholesteric reflected colour of these combinations for a rewritable colour image recording. The (E)-azobenzenes (~2 wt%) in the host CLC induced pitch changes dependent on temperature, the concentration of the azobenzenes and the length of substituted alkyl chain on the azobenzenes. In the low region of the cholesteric temperature (below 100 °C), some (E)-azobenzenes remarkably lengthened the pitch. Contrary to this, upon irradiation, E–Z photoisomerization of all the azobenzenes in the CLC phase induced consecutive pitch shortenings in response to the formation of the Z isomers. The changed pitch (reflected colour) could be fixed in cholesteric glassy solids. In the glassy solid state, the photo and thermal isomerization of the azobenzenes could not influence the cholesteric pitch any longer, therefore, the colour was stably stored. However, since the molecular arrangement of the CLC and the conformation of the doped azobenzenes completely returned to the initial state upon re-heating to isotropic temperatures, the stored colour could be easily erased. Based on these fundamental investigations, it is suggested that the mixed systems of the medium-molecular-weight CLCs and the 4,4′-dialkylazobenzenes will be effective materials for a rewritable full-colour image recording in a photon mode.

 Please wait while we load your content...
Please wait while we load your content...