Mechanistic insights into perovskite photoluminescence enhancement: light curing with oxygen can boost yield thousandfold†
Abstract
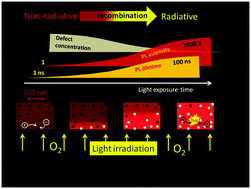
A light-induced photoluminescence (PL) enhancement in surface-deposited methylammonium lead iodide (CH3NH3PbI3) perovskites was investigated in detail using time-resolved luminescence microscopy. We found the PL intensity to increase up to three orders of magnitude upon light illumination with an excitation power density of 0.01–1 W cm−2. The PL enhancement is accompanied by an increase of the PL lifetime from several nanoseconds to several hundred nanoseconds and also by an increase of the initial amplitude of the PL decay. The latter suggests excited state quenching at the subpicosecond timescale. We propose a model where the trapping sites responsible for non-radiative charge recombination can be de-activated by a photochemical reaction involving oxygen. The reaction zone is spatially limited by the excitation light-penetration depth and diffusion length of the charge carriers. The latter increases in the course of the light-curing process making the reaction zone spreading from the surface towards the interior of the crystal. The PL enhancement can be reversed by switching on/off the excitation light or switching the atmosphere between oxygen and nitrogen. Slow diffusion of the reactants and products and equilibrium between the active and “cured” trapping sites are proposed to be the reasons for peculiar responses of PL to such varied experimental conditions.


 Please wait while we load your content...
Please wait while we load your content...