Organocatalytic enantioselective synthesis of β-amino sulfonic acid derivatives†
Abstract
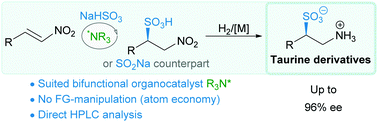
An unprecedented enantioselective conjugate addition reaction of sodium bisulfite to various nitrostyrenes occurred upon the influence of a bifunctional amino-thiourea organocatalyst; a strategy that opens a straightforward route to unprotected chiral taurine derivatives thanks to the reduction of the obtained β-nitroethanesulfonic acids into the corresponding amino derivatives.


 Please wait while we load your content...
Please wait while we load your content...