Bimetallic Co2Mo3O8 suboxides coupled with conductive cobalt nanowires for efficient and durable hydrogen evolution in alkaline electrolyte†
Abstract
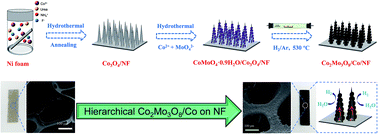
Development of highly active and robust earth-abundant electrocatalysts for the hydrogen evolution reaction (HER) is of great significance for the broad utilization of alkaline electrolyzers. Herein, cobalt molybdenum suboxides (Co2Mo3O8) supported on Ni foam are reduced from their corresponding oxide precursors and first used as efficient electrocatalysts for the HER in alkaline electrolyte. The catalytic activity is further improved through the construction of a unique hierarchical Co2Mo3O8/Co/NF nanostructure. With a very low onset overpotential (∼9 mV), this electrocatalyst only requires overpotentials of 50 mV and 137 mV to drive current densities of 10 mA cm−2 and 100 mA cm−2, respectively. After 5000 cycles, a slight increase of ∼6 mV in the overpotential to drive a current density of 10 mA cm−2 verifies the excellent stability of Co2Mo3O8/Co/NF during the alkaline HER process.


 Please wait while we load your content...
Please wait while we load your content...