Sustainable chromic acid oxidation: solar-driven recycling of hexavalent chromium ions for quinone production by WO3 nanosponge photoanodes†
Abstract
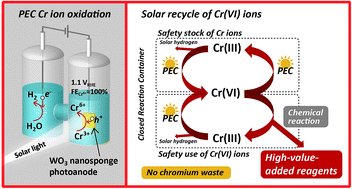
Photoelectrochemical chromium ion oxidation from the trivalent to the hexavalent state has been demonstrated by using WO3 nanosponge photoanodes in sulfuric acid electrolytes under simulated sunlight. Faradaic efficiencies of almost 100% were achieved in electrolytes containing high Cr3+ (≥10 mM) and low SO42− (≤10 mM) concentrations at very low bias potential voltages below 1.1 VRHE. At the photoanode, the production of Cr6+ ions competed with the formation of S2O82− ions from HSO4−/SO42− ions. At the cathode, hydrogen evolution occurred with 100% faradaic efficiency. Additionally, Cr3+ recovery was also observed at the cathode upon switching the electrolytes of the anode and cathode cells after the Cr6+ production, demonstrating the complete cyclic photoelectrochemical reaction between Cr3+ and Cr6+ ions. Finally, we showed the applicability of chromic acid oxidation to quinone production by using the photoelectrochemically produced Cr6+ ions in the sulfuric acid solutions. p-Benzenediol was successfully converted into p-benzoquinone with high efficiency. Thus, we have demonstrated the production of high-value-added organic reagents via photoelectrochemically driven chromic acid oxidation with the recycling of Cr6+ from Cr3+ using solar energy.
- This article is part of the themed collection: 2018 Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...