Facile fabrication of a 3D network composed of N-doped carbon-coated core–shell metal oxides/phosphides for highly efficient water splitting†
Abstract
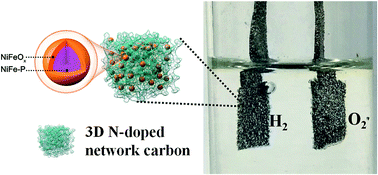
Development of robust, bifunctional, and non-precious catalysts for oxygen and hydrogen evolution reactions (OER and HER) is a prerequisite to realizing the overall splitting of water. This, however, remains a great challenge. In this context, we fabricated a novel three-dimensional (3D) network comprising N-doped carbon-coated core–shell NiFeOx@NiFe–P (denoted as NC–NiFeOx@NiFe–P) by two-pot high-temperature phosphorization and surface oxidation of a NiFe-Prussian blue analogue/polyvinylpyrrolidone (denoted as NiFe–PBAs/PVP) hybrid precursor. The as-synthesized NC–NiFeOx@NiFe–P catalyst demonstrated exceptional performance for both OER and HER, offering a current density of 10 mA cm−2 (a metric related to solar fuel) at small overpotentials of 285 mV for the OER and 237 mV for the HER in 1 M KOH, respectively. As expected, a NC–NiFeOx@NiFe–P based alkaline electrolyzer with durability of 20 h was manufactured to achieve 10 mA cm−2 at a voltage of 1.59 V, outperforming most non-precious metal-based electrolyzers. The exceptional performance could be attributed to the unique 3D network composed of core–shell NiFeOx@NiFe–P and highly conductive N-doped carbon (NC), which provided a large amount of highly active sites for both OER and HER and favored fast electron transport during electrocatalytic processes.


 Please wait while we load your content...
Please wait while we load your content...