Electronic modulation of transition metal phosphide via doping as efficient and pH-universal electrocatalysts for hydrogen evolution reaction†
Abstract
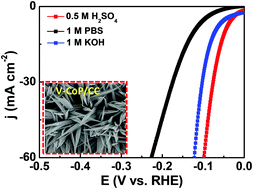
It is highly desirable to develop efficient and low-cost catalysts to minimize the overpotential of the hydrogen evolution reaction (HER) for large-scale hydrogen production from electrochemical water splitting. Doping a foreign element into the host catalysts has been proposed as an effective approach to optimize the electronic characteristics of catalysts and thus improve their electrocatalytic performance. Herein we, for the first time, report vanadium-doped CoP on self-supported conductive carbon cloth (V-CoP/CC) as a robust HER electrocatalyst, which achieves ultra-low overpotentials of 71, 123 and 47 mV to afford a current density of 10 mA cm−2 in 1 M KOH, 1 M PBS and 0.5 M H2SO4 media, respectively. Meanwhile, the V-CoP/CC electrode exhibits a small Tafel slope and superior long-term stability over a wide pH range. Detailed characterizations reveal that the modulation of the electronic structure contributes to the superior HER performance of V-CoP/CC. We believe that doping engineering opens up new opportunities to improve the HER catalytic activity of transition metal phosphides through regulating their physicochemical and electrochemical properties.
- This article is part of the themed collection: Most popular 2018-2019 catalysis articles


 Please wait while we load your content...
Please wait while we load your content...