Thermal and photo-RAFT polymerization of 2,2,2-trifluoroethyl α-fluoroacrylate†
Abstract
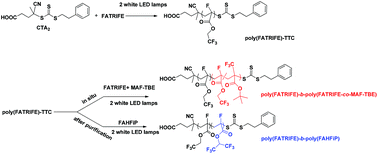
The RAFT polymerization of 2,2,2-trifluoroethyl α-fluoroacrylate (FATRIFE) was investigated under thermal conditions and light irradiation. The performances of 4 different chain transfer agents (3,5-dimethyl-1H-pyrazole-1-carbodithioate (CTA1), 4-cyano-4-(2-phenylethanesulfanylthiocarbonyl)sulfanylpentanoic acid (CTA2), dibenzyl trithiocarbonate (CTA3), and ethyl 2-(phenylcarbonothioylthio) propionate (CTA4)) were compared under thermal conditions. CTA2 afforded relatively good control of the polymerization with relatively low dispersities (apparent molar mass Mn,SEC ∼ 37.8 kg mol−1vs. PMMA, Đ < 1.10). Pseudo-first-order polymerization kinetics and a linear increase of the molar masses versus monomer conversions were observed. The RAFT polymerization of FATRIFE was further explored under photoirradiation using CTA2 as the chain transfer agent and white LED lamps (14 W × 2). In the absence of exogenous radical sources or catalysts, controlled polymerization was observed at room temperature. A linear increase of molar masses (up to 25.3 kg mol−1) with monomer conversions and low dispersities (Đ < 1.10) were successfully obtained. Temporal control of the polymerization was also observed using light ON/OFF experiments. Poly(FATRIFE) based macroCTA synthesized by photoRAFT was also extended with 1,1,1,3,3,3-hexafluoropropan-2-yl 2-fluoroacrylate and tert-butyl-2-trifluoromethacrylate/FATRIFE.


 Please wait while we load your content...
Please wait while we load your content...