Synthesis and characterization of ureido-derivatized UCST-type poly(ionic liquid) microgels†
Abstract
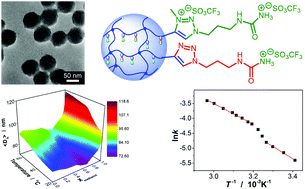
We report a class of stimuli-responsive poly(ionic liquid) microgel that possesses an upper critical solution temperature (UCST). Such a microgel is synthesized from ureido polymers via quaternary ammonization and post synthetic modification by anion-exchange treatment. These microgels can undergo reversible UCST-type volume phase transitions in response to the changes in temperature of water, methanol, and water/methanol mixtures, such that the temperature-responsive volume phase transition behavior can be readily tuned over a significantly wide range in a miscible mixture of water/methanol by varying the mol ratio of the two solvents, resulting in a phase diagram displaying the UCST-type co-nonsolvency phase separation phenomenon. At a set temperature in our experimental temperature window of 20.0–64.0 °C, the microgels can undergo an isothermal re-entrant swelling–shrinking–swelling transition as the methanol content increases in the water/methanol mixture. Moreover, these microgels as catalysts in a model esterification reaction of palmitic acid with methanol can display merits of both homogeneous (considerably high efficient catalytic activity) and heterogeneous (excellent recyclability) catalysis. With UCST-type responsive phase behavior and catalytic properties harnessed on the same object, the microgels react to external stimuli which allows their catalytic properties to be altered accordingly, to a certain extent, in a novel non-monotonous way different from that reported previously on the catalyst systems using LCST-type microgels.


 Please wait while we load your content...
Please wait while we load your content...