Metal ion assisted interface re-engineering of a ferritin nanocage for enhanced biofunctions and cancer therapy†
Abstract
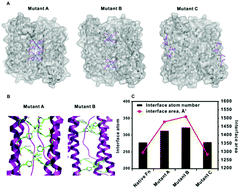
The bottom-up self-assembly of protein subunits into supramolecular nanoarchitectures is ubiquitously exploited to recapitulate and expand the features of natural proteins to advance nanoscience in medicine. Various chemical and biological re-engineering approaches are available to render diverse functions in the given proteins. They are, unfortunately, capable of compromising protein integrity and stability after extensive modifications. In this study, we introduce a new protein re-engineering method, metal ion assisted interface re-engineering (MAIR), to serve as a robust and universal strategy to extend the functions of self-assembly proteins by boosting structural features to advance their diverse biomedical applications. In particular, the MAIR strategy was applied to a widely used natural protein, ferritin, as a model protein to coordinate with copper ions in its mutagenic artificial metal binding domain. Structure directed rational protein mutagenesis was carried out at the C2 interface amino acid residues of the ferritin subunit for metal ion coordination site optimization. Copper binding at the artificial binding pocket was highly specific over the other divalent ions present in physiological fluids, and the structurally embedded copper ion in turn strengthened the overall protein integrity and stability. In the presence of isotopic copper-64, the interface re-engineered ferritin worked as a chelator-free molecular nanoprobe with an extraordinarily high specific activity to allow PET imaging of tumors in live animals. We also found that the re-engineered ferritin coordinating with copper ions demonstrates high drug loading capacity of a widely used anti-cancer agent, doxorubicin (DOX), to achieve significant drug retention at the tumor site and enhance tumor regression for improved anti-cancer effects. The MAIR approach, thus, exploited the copper ion to facilitate efficient one-step labeling of mutant ferritin derivatives for simultaneous molecular imaging and drug delivery. The reported interface re-engineering strategy provides an unparalleled opportunity to expand protein biofunctions to serve as a new theranostic agent in cancer research.
- This article is part of the themed collection: Nanoscale 10th Anniversary: Top Authors


 Please wait while we load your content...
Please wait while we load your content...