New oxidovanadium(iv) complex with a BIAN ligand: synthesis, structure, redox properties and catalytic activity†
Abstract
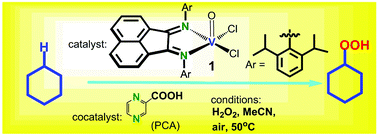
Reaction of VCl3 with bis[N-(2,6-diisopropylphenyl)imino]acenaphthene (dpp-bian) in air afforded [VOCl2(dpp-bian)] (1). The complex was characterized by IR and UV-vis spectroscopies and elemental analysis. The crystal structure of 1 was determined by X-ray diffraction (XRD) analysis. The vanadium atom is in a square-pyramidal OCl2N4 coordination environment. The cyclic voltammogram (CV) in dichloromethane reveals an irreversible oxidation process at +1.40 V (vs. Ag/AgCl) assigned to the V(IV)/V(V) couple, and two consecutive quasi-reversible one-electron reduction processes at −0.32 V and −1.05 V (vs. Ag/AgCl), respectively, assigned to the bian/bian−/˙ and bian−/˙/bian2− couples, followed by irreversible reduction at −1.6 V (vs. Ag/AgCl). The EPR spectrum of 1 in toluene shows a single 8-line signal typical for oxidovanadium(IV) complexes with d1 configuration. The magnetic behavior of 1 confirms the presence of one unpaired electron (μeff (330 K) = 1.67 μB), and the isolation of the paramagnetic centers. Application of 1 to oxidation of alkanes documented high catalytic activity under mild conditions. The kinetics and selectivity of alkane oxygenation by the 1/H2O2 and 1/PCA/H2O2 systems (PCA is pyrazine-2-carboxylic acid) were studied. The reaction is more efficient in the presence of PCA.


 Please wait while we load your content...
Please wait while we load your content...