A turn-on fluorescence assay of alkaline phosphatase activity using a DNA–silver nanocluster probe†
Abstract
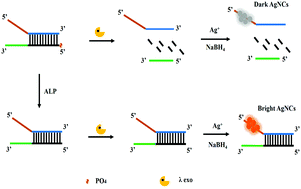
Assays of alkaline phosphatase (ALP) activity play a critical role in clinical diagnostics and drug screening. In this work, we present a novel, sensitive and cost-effective analytical method for the detection of ALP activity based on the design of a G-rich DNA light-up DNA–silver nanocluster (AgNCs) probe and λ exonuclease (exo) cleavage reaction. Upon addition of λ exo, the G-rich DNA was cleaved, resulting in the inhibition of AgNCs to move closer to G-rich DNA sequences and, accordingly, only a low fluorescence signal was observed. Upon treatment of ALP, the 5′-phosphoryl end of pG-rich DNA was hydrolyzed and the λ exo cleavage reaction was impeded. The AgNCs were then enabled to move closer to the G-rich DNA sequences, resulting in an increase in fluorescence. Under optimized conditions, the fluorescence change was determined to be linear with the ALP concentration ranging between 1 U L−1 and 800 U L−1 (detection limit: 1 U L−1). Taken in concert, this strategy may provide a basis for a screening platform for ALP inhibitors.


 Please wait while we load your content...
Please wait while we load your content...