Synthesis of TiO2@α-Fe2O3 core–shell heteronanostructures by thermal decomposition approach and their application towards sunlight-driven photodegradation of rhodamine B†
Abstract
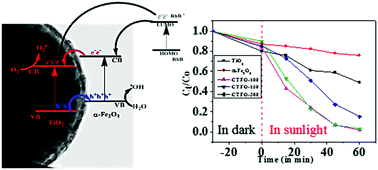
TiO2@α-Fe2O3 core–shell heteronanostructures with different shell thickness have been synthesized by a thermal decomposition approach. The thickness of the shell (α-Fe2O3) could be controlled by simply varying the amount of iron–urea complex, which was used as a precursor for iron oxide. The formation of core–shell TiO2@α-Fe2O3 was confirmed by various analytical techniques. XRD analysis confirms the presence of anatase and hematite in the TiO2@α-Fe2O3 core–shell heteronanostructures. Scanning electron microscopy studies confirm deposition of the α-Fe2O3 shell on the surface of TiO2 spheres and TEM analysis proves the formation of a uniform α-Fe2O3 shell on the TiO2 spheres. The XPS measurements prove the presence of Ti4+, Fe3+, and oxygen in the lattice and surface hydroxyls. From UV-visible DRS spectroscopy, it was observed that the TiO2@α-Fe2O3 core–shell heteronanostructures absorb in both the UV and visible range of the electromagnetic spectrum. Field-dependent (M–H) magnetic measurements indicate the weak ferromagnetic behaviour of the TiO2@α-Fe2O3 core–shell nanoparticles at 300 K and superparamagnetic behaviour at 5 K. Temperature-dependent magnetic studies (M–T) show the characteristic Morin transition of α-Fe2O3 in all the core–shell nanoparticles. The synthesized TiO2@α-Fe2O3 core–shell heteronanostructures were explored as a catalyst for the photodegradation of rhodamine B (RhB) in an aqueous solution in the presence of sunlight and it was found that TiO2@α-Fe2O3 core–shell nanoparticles act as a better photocatalyst compared to pure TiO2 and α-Fe2O3. Kinetic studies indicate that the photodegradation follows first-order kinetics.


 Please wait while we load your content...
Please wait while we load your content...