Synthesis of novel AIEE active pyridopyrazines and their applications as chromogenic and fluorogenic probes for Hg2+ detection in aqueous media†‡
Abstract
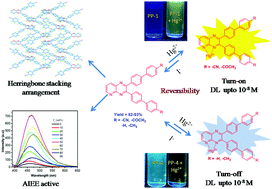
A series of novel pyridopyrazine derivatives was synthesized in excellent yields. All compounds displayed weak fluorescence emission in solution but showed strong emission in the aggregated state indicating aggregation induced emission enhancement (AIEE) characteristics. Single crystal analysis of PP1 showed a highly twisted conformation with no π–π stacking interactions. The application of these pyridoyrazines as chromogenic and fluorogenic probes for the detection of Hg2+ ions is also reported. Pyridopyrazine derivatives bearing electron-withdrawing biphenyl rings (PP1 and PP2) act as excellent “turn-on” fluorescent probes while those bearing electron-donating biphenyl rings (PP3 and PP4) act as excellent “turn-off” fluorescent probes for Hg2+ ions in non-buffered aqueous media with high selectivity in the presence of competitive metal ions such as Na+, K+, Mg2+, Ca2+, Ba2+, Cr3+, Fe3+, Fe2+, Co2+, Ni2+, Cu2+, Ag+, Zn2+, Cd2+, Al3+ and Pb2+. A visual color change from colorless to yellow upon treatment with Hg2+ was also observed for all probes; thus, they could also be used for naked eye detection of Hg2+ ions. These probes displayed selective detection of Hg2+ ions in the range from 3.37 × 10−7 M to 8.17 × 10−8 M. The 1 : 1 binding stoichiometry for the complex formation between the pyridopyrazine derivative and Hg2+ was confirmed by Job's plot and NMR studies. Binding of Hg2+ to pyridopyrazines was found to be reversible upon addition of KI and the process could be repeated several times. Additionally, filter paper strips coated with PP1 and PP2 were also used for the detection of Hg2+ ions.


 Please wait while we load your content...
Please wait while we load your content...