Theoretical study on the interaction of CO2 and H2O molecules with metal doped-fluorinated phthalocyanines†
Abstract
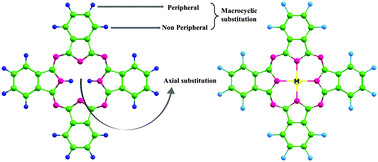
This investigation reports the molecular properties of metal (M = Mg, Sc, Cu, Zn) doped-fluorinated phthalocyanines (M-FPcs) and their ability to separate and capture CO2 in the presence of H2O using DFT studies. The structural investigations of M-FPcs show that Sc-FPc alone is non-planar while other M-FPcs exhibit a planar geometry. The HOMO–LUMO gap shows that Sc-FPc is least stable and hence more reactive. The DOS plot shows the influence of 3d-orbitals of Sc, Cu and Zn on F-Pc. On interacting M-FPcs with CO2 and H2O, it can be seen that the interaction of H2O is greater than CO2. The trend of CO2 interaction is as follows: Sc > Mg > Zn > Cu. Furthermore, Gibbs free energy for the Sc-FPc + CO2 system shows that the interaction is spontaneous and feasible while for Mg, Cu and Zn FPc the interaction is non-spontaneous. However, H2O spontaneously interacts with all the metal FPcs and destabilizes the systems except for Sc-FPc in which H2O interaction stabilizes the molecule. The calculated adsorption capacity (n) value shows that all the metal–phthalocyanine systems have higher adsorption capacity for CO2 than for H2O. Interestingly, the adsorption of CO2 on Sc-FPc differs by an order of 108 with respect to H2O. Furthermore, the adsorption capacity is low for systems having metals with high van der Waals radii. Conclusively, the Sc-FPc is the best-suited material for CO2 separation and capture in the presence and absence of water molecules, respectively.


 Please wait while we load your content...
Please wait while we load your content...