The effect of alkyl chain tethers on the kinetics and mechanistic behaviour of bifunctional dinuclear platinum(ii) complexes bearing N,N′-dipyridylamine ligands†
Abstract
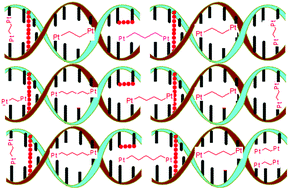
In the current paper, we report the kinetics of bifunctional dinuclear platinum(II) complexes viz., 1,2-N,N′-di-(2,2-dipyridylamine)ethanetetraaquaplatinum(II), PtL2, 1,3-N,N′-di-(2,2-dipyridylamine)propanetetraaquaplatinum(II), PtL3, 1,4-N,N′-di-(2,2-dipyridylamine)butanetetraaquaplatinum(II), PtL4, 1,5-N,N′-di-(2,2-dipyridylamine)pentanetetraaquaplatinum(II), PtL5 and 1,6-N,N′-di-(2,2-dipyridylamine)hexanetetraaquaplatinum(II), PtL6. The substitution reactions were carried out on tetraaqua complexes with thiourea nucleophiles under pseudo-first-order conditions as a function of nucleophile concentration and temperature by stopped-flow and UV-vis spectrophotometric techniques. An experimental study was conducted with the aim of determining the influence of alkyl chains on the steric and electronic structure of dinuclear platinum(II) complexes. The reactivity of these complexes was dependent on the length of the alkyl spacer. The results obtained herein demonstrate the intriguing odd–even effects induced by the alkyl chain on the complexes. Artificial constraints imposed by the alkyl chain significantly affect their conformational structure to be either synperiplanar (syn-) or antiperiplanar (anti-) characterized by the odd and even effect. The kinetic, mechanistic and conformational behaviour was influenced by the size of the alkyl chain in accordance with odd–even alterations of the spacer. Computational modeling using density functional theory (DFT) calculations supplemented experimental findings that structural features and the reactivity pattern of these organometallic complexes are governed by both steric and electronic effects arising from the flexibility and inductive nature of the alkyl spacer. The strong σ-donicity of longer alkyl chains favours sufficient accumulation of electron density at the metal centre and stabilizes a 14-electron intermediate. The study shows the HOMO–LUMO energy (ΔE) is affected by the length of the spacer. Kinetic and DFT data indicate electron donation by the alkyl spacer. The low positive values of enthalpy of activation and significantly large negative values of entropy of activation indicate an associative mechanism of substitution.


 Please wait while we load your content...
Please wait while we load your content...