Exploring eukaryotic versus prokaryotic ribosomal RNA recognition with aminoglycoside derivatives†
Abstract
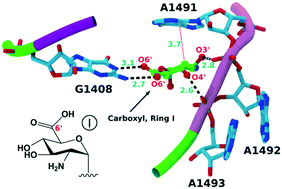
New derivatives of aminoglycosides containing 6′-carboxylic acid or 6′-amide on their ring I were designed, synthesized and their ability to readthrough nonsense mutations was examined in vitro, along with the protein translation inhibition in prokaryotic and eukaryotic systems. The observed structure–activity relationships, along with the comparative molecular dynamics simulations within the eukaryotic rRNA decoding site, showed high sensitivity of 6′-position to substitution, indicating that the rational design of potent stop-codon read-through inducers requires consideration of not only the structure and energetics of the drug–RNA interaction but also the dynamics associated with that interaction.


 Please wait while we load your content...
Please wait while we load your content...