A loop engineering strategy improves laccase lcc2 activity in ionic liquid and aqueous solution†
Abstract
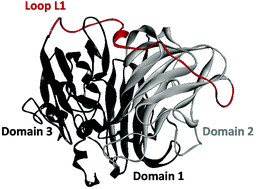
Laccases, especially high redox potential laccases, play an important role in lignin degradation. The fungal laccase lcc2 from Trametes versicolor has a high redox potential and EMIM- and BMIM-based ionic liquids show excellent solubilization of wood-derived biomass. Concentrations of EMIM and BMIM to efficiently dissolve lignin impede laccase activity. Protein engineering to improve the activity and resistance of laccases in ionic liquids offers a promising opportunity for lignin valorization for the sustainable production of fuels and bulk high-value chemicals. In this work, we have performed computational assisted protein engineering of lcc2 to increase its performance in the presence of ionic liquid and aqueous solution. We showed that the loop L1 (amino acid residues 284–320) is highly important for improving lcc2 activity in EMIM EtSO4 and aqueous solutions. Lcc2 activity was improved based on a KnowVolution campaign through site saturation of seven amino acid positions identified by computational modeling. Simultaneous site saturation of four amino acid positions by OmniChange yielded variants OM1 (A285P/A310R/A312E/A318G) and OM3 (A310D/A312P/A318R) with a 3.9-fold (535.8 ± 36.9 U mg−1) and 1.6-fold (216.8 ± 5.3 U mg−1) increased specific activity in aqueous solution (lcc2 WT, 138.9 ± 6.5 U mg−1), respectively. High conservation of the loop L1 in fungal laccases suggests that computational assisted loop engineering might be used as a general strategy to improve their activity in ionic liquids and aqueous solutions.


 Please wait while we load your content...
Please wait while we load your content...