Degradation of 1H-benzotriazole by UV/H2O2 and UV/TiO2: kinetics, mechanisms, products and toxicology†
Abstract
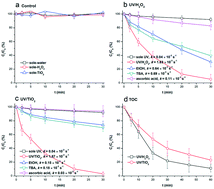
Benzotriazoles are emerging contaminants widespread in environmental waters. As they are robust against conventional biological wastewater treatment, it is desirable to develop cost-effective and safe treatment methods for benzotriazole removal. The current study attempted to investigate the degradation of water dissolved 1H-benzotriazole (1H-BTA) with UV/H2O2 and UV/TiO2. Pseudo-first order degradation kinetics were observed in low power 280 nm UV/H2O2 and UV/TiO2 systems (UV intensity = 0.023 mW cm−2, kapp reached 1.63 × 10−3 s−1 and 1.87 × 10−3 s−1, respectively), and radical oxidation was the dominant reaction mechanism with k˙OH-BTA at (7.1 ± 0.8) × 109 M−1 s−1 and (6.9 ± 0.7) × 109 M−1 s−1. Both systems were affected by the pH value, natural organic matter and anions, leading to incomplete mineralization in actual water treatment processes. As the reaction proceeded, 1H-BTA was progressively transformed into eight organic products. The number of preliminary hydroxylated products (e.g. C6H5N3O) increased rapidly at the early stage, while the further open-loop products (e.g. C4H3N3O4) were dominant at the later stage. Based on the proteomics analysis, the significant activation of ribosome, transporter and tricarboxylic acid cycle metabolisms in Escherichia coli, which exposed to the later degradation product mixture, suggested that the toxicity of 1H-BTA decreased. In conclusion, incomplete mineralization using hydroxyl radical oxidation likewise has potential for thedetoxification of 1H-BTA.
- This article is part of the themed collection: Ultraviolet-based Advanced Oxidation Processes (UV AOPs)


 Please wait while we load your content...
Please wait while we load your content...