Aqueous vs. nonaqueous Zn-ion batteries: consequences of the desolvation penalty at the interface†
Abstract
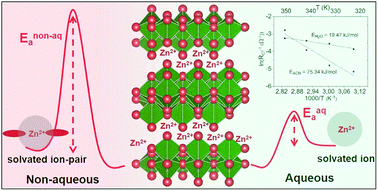
Zinc ion batteries using metallic zinc as the negative electrode have gained considerable interest for electrochemical energy storage, whose development is crucial for the adoption of renewable energy technologies, as zinc has a very high volumetric capacity (5845 mA h cm−3), is inexpensive and compatible with aqueous electrolytes. However, the divalent charge of zinc ions, which restricts the choice of host material due to hindered solid-state diffusion, can also pose a problem for interfacial charge transfer. Here, we report our findings on reversible intercalation of up to two Zn2+ ions in layered V3O7·H2O. This material exhibits very high capacity and power (375 mA h g−1 at a 1C rate, and 275 mA h g−1 at an 8C rate) in an aqueous electrolyte compared to a very low capacity and slow rate capabilities in a nonaqueous medium. Operando XRD studies, together with impedance analysis, reveal solid solution behavior associated with Zn2+-ion diffusion within a water monolayer in the interlayer gap in both systems, but very sluggish interfacial charge transfer in the nonaqueous electrolyte. This points to desolvation at the interface as a major factor in dictating the kinetics. Temperature dependent impedance studies show high activation energies associated with the nonaqueous charge transfer process, identifying the origin of poor electrochemical performance.
- This article is part of the themed collection: Celebrating Excellence in Research: 100 Women of Chemistry


 Please wait while we load your content...
Please wait while we load your content...