Enrichment of rare earth metal ions by the highly selective adsorption of phytate intercalated layered double hydroxide
Abstract
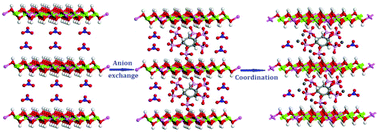
Phytate intercalated MgAl layered double hydroxide (MgAl-LDH) was prepared by an anion exchange method with the precursor NO3− containing MgAl-LDH. The final as-synthesized product [Mg0.69Al0.31(OH)2] (phytateNa6)0.05 (NO3)0.01·mH2O (phytate-LDH) has highly selective adsorption ability for some metal ions and can be used to enrich rare earth metal ions in mixed solution, such as Pr3+ and Ce3+ from a mixed solution of them with Pb2+ and Co2+. At first, phytate-LDH has good adsorption performance for these ions in single metal ion solutions. At low concentration (below 10 mg L−1), all the capture rates of the four metal ions were more than 97%, for highly toxic Pb2+ it was even up to nearly 100%, and a high capture rate (99.87%) was maintained for Pb2+ at a high concentration (100 mg L−1). When all the four metal ions are co-existing in aqueous solution, the selectivity order is Pb2+ ≫ Pr3+ ≈ Ce3+ > Co2+. In a solution containing mixtures of the three metal ions of Pr3+, Ce3+, and Co2+, the selectivity order is Pr3+ ≈ Ce3+ ≫ Co2+, and in a solution containing mixtures of Pr3+ with Co2+ and Ce3+ with Co2+, the selectivity orders are Pr3+ ≫ Co2+ and Ce3+ ≫ Co2+, respectively. The high selectivity and adsorption capacities for Pb2+, Co2+, Pr3+, and Ce3+ result in the efficient removal of Pb2+ and enrichment of the rare earth metal ions Pr3+ and Ce3+ by phytate-LDH. Based on the elemental analysis, it is found that the difference of the adsorption capacities is mainly due to the different coordination number of them with phytate-LDH. With molecular simulation, we believe that the adsorption selectivity is due to the difference of the binding energy between the metal ion and phytate-LDH. Therefore, the phytate-LDH is promising for the enrichment and/or purification of the rare earth metal ions and removal of toxic metal ions from waste water.
- This article is part of the themed collection: Recent Developments in Intercalation Compounds: Chemistry and Applications


 Please wait while we load your content...
Please wait while we load your content...