Kinetic analysis of hexose conversion to methyl lactate by Sn-Beta: effects of substrate masking and of water†
Abstract
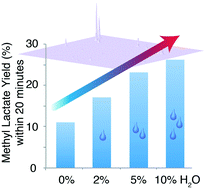
Simple sugars show promise as substrates for the formation of fuels and chemicals using heterogeneous catalysts in alcoholic solvents. Sn-Beta is a particularly well-suited catalyst for the cleavage, isomerization and dehydration of sugars into more valuable chemicals. In order to understand these processes and save resources and time by optimising them, kinetic and mechanistic analyses are helpful. Herein, we study substrate entry into the Sn-Beta-catalysed methyl lactate process using abundant hexose substrates. NMR spectroscopy is applied to show that the formation of methyl lactate occurs in two kinetic regimes for fructose, glucose and sucrose. The majority of methyl lactate is not formed from the substrate directly, but from methyl fructosides in a slow regime. At 160 °C, more than 40% of substrate carbon are masked (i.e. reversibly protected in situ) as methyl fructosides within a few minutes when using hydrothermally synthesised Sn-Beta, while more than 60% methyl fructosides can be produced within a few minutes using post-synthetically treated Sn-Beta. A significant fraction of the substrate is thus masked by rapid methyl fructoside formation prior to subsequent slow release of fructose. This release is the rate-limiting step in the Sn-Beta-catalysed methyl lactate process, but it can be accelerated by the addition of small amounts of water at the expense of the maximum methyl lactate yield.


 Please wait while we load your content...
Please wait while we load your content...