Cu–Mg–Zr/SiO2 catalyst for the selective hydrogenation of ethylene carbonate to methanol and ethylene glycol†
Abstract
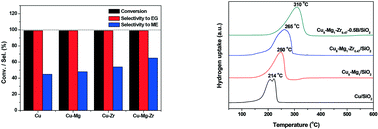
A Cux–Mgy–Zrz/SiO2 catalyst with a total metal loading of 60 wt% prepared by a deposition–precipitation method was applied for the selective hydrogenation of ethylene carbonate to methanol and ethylene glycol in a fixed-bed reactor. As a result, the Cu8–Mg1–Zr0.47/SiO2 catalyst furnished 99% ethylene carbonate conversion with 85% selectivity to methanol and 99% selectivity to ethylene glycol under the optimized reaction conditions. Moreover, the Cu8–Mg1–Zr0.47/SiO2 catalyst also showed a good lifetime and neither the activity nor the selectivity decreased the during 208 h test. The reaction was found to depend sensitively on the Cu particle size, the surface acidity and the catalyst surface composition. The synergistic effect of balanced Cu0 and Cu+ sites was considered to play a critical role in attaining high yields of methanol and ethylene glycol. Boric oxide also had a positive effect on the hydrogenation of ethylene carbonate, affording higher selectivity to methanol under much milder conditions.


 Please wait while we load your content...
Please wait while we load your content...