Understanding the activity and selectivity of single atom catalysts for hydrogen and oxygen evolution via ab initial study†
Abstract
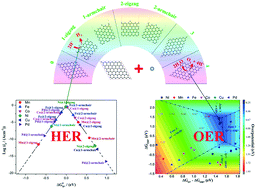
To identify the specific activity sites for both the HER and OER in experimental realized single transition-metal atom decorated graphene sheets, we assume the number of metal–C bonds (coordination) determines the adsorption strength of reaction intermediates on the metal atom sites. We find the strength weakens with the metal coordination number. Low-coordinated Ni sites, i.e. single-coordinated Ni on the zigzag edge, exhibit the highest activity toward HER, while high-coordinated Ni atoms, i.e. quadruple-coordinated Ni, demonstrate the best OER performance. This assumption has been further confirmed by a wide range of transition metals, including Mn, Fe, Co, Cu, and Pd. Our findings highlight a new family of efficient bifunctional catalysts for water splitting and offer a new paradigm for the design of single-atom HER/OER catalysts toward hydrogen production.


 Please wait while we load your content...
Please wait while we load your content...