Selective hydrogenation of CO2 to methanol catalyzed by Cu supported on rod-like La2O2CO3†
Abstract
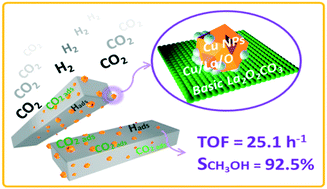
Cu-based catalysts have long been applied to convert CO2 and H2 into methanol, and their performances are well known to be markedly influenced by the support and promoter. Herein, several Cu-based catalysts supported on lanthanum oxides were fabricated by varying the preparation methods and characterized by XRD, TEM, ICP-AES, N2 physisorption, N2O chemisorption, CO2 chemisorption, XPS, and CO2-TPD. The results showed that the as-prepared Cu supported on rod-like La2O2CO3 (La2O2CO3-R) exhibited the highest TOFCu, methanol selectivity and yields of methanol for the hydrogenation of CO2 to methanol. Distinct from conventional promoter addition, the local formation of a new type of Cuδ+ species at Cu/La2O2CO3-R interfaces and the original adsorption performance of basic oxides are the two key factors relating to the catalytic performance. Cu supported on La2O2CO3-R with a higher content of Cuδ+ species and a stronger adsorption for CO2 leads to superior catalytic activities. DRIFTS studies revealed that the generation of synergetic basic sites at the interfacial areas can increase the intrinsic activity of Cu-based methanol catalysts by moderately and selectively stabilizing methanol synthesis intermediates. This work provides new insights into CO2 activation over basic oxide-supported Cu catalysts, and the identification of metal–support interactions between Cu and lanthanum oxides is beneficial for the rational design of stable Cu-based catalysts.


 Please wait while we load your content...
Please wait while we load your content...