Effect of halogen dopants on the properties of Li2O2: is chloride special?†
Abstract
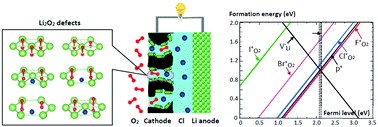
There is consensus on the fact that one of the main limitations of Li air batteries (LABs) is the insulating character of Li2O2 and that it becomes crucial to explore new conduction paths. Recent studies indicate that doping with chloride increases the ion conductivity of Li2O2, although to a much lesser extent than expected if chloride is assumed to be a donor dopant [Gerbig et al., Adv. Mater., 2013, 25, 3129]. Subsequently, it has been shown that the addition of lithium chloride, LiCl, to the battery electrolyte increases its discharge capacity, while this effect is not observed with other halogens [Matsuda et al., J. Phys. Chem. C, 2016, 120, 13360]. This fact was attributed to an increase in the conductivity of Cl-doped Li2O2, but still the responsible mechanism is not clear. In this work, we have performed first principle calculations to study the effect of the different halogens (F, Cl, Br, I) as substitutional defects on the electronic and transport properties of Li2O2. We have calculated the formation energies of the different defects and impurities and we analysed how they affect the activation barriers and diffusion coefficients. We have demonstrated that the chloride does not behave like a donor dopant, thus explaining the meager increase of the ionic conductivity experimentally observed, and neither does it promote polaron formation and mobility. We have also found that chloride does not present any special behaviour among the halogen series. Our results reveal that all the studied configurations associated with the halogen defects do not derive metallic states nor extra polarons that would increase considerably the electronic conductivity. This is mainly due to the ionic characteristics of the Li2O2 crystal and the capability of the oxygen dimers to adapt its valence rather than to the nature of the dopant itself.


 Please wait while we load your content...
Please wait while we load your content...