The influence of a hydrophobic carrier, reactant and product during H2O adsorption on Pd surface for the oxidative esterification of methacrolein to methyl methacrylate
Abstract
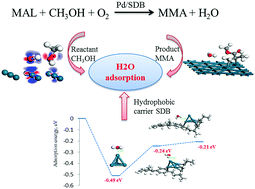
Taking the one-step oxidative esterification of methacrolein (MAL) to methyl methacrylate (MMA) as a model reaction and because H2O that was generated easily formed a film of water on the catalyst surface, which restricted the diffusion of the reactants to the active sites, the effects of the hydrophobic carrier styrene–divinylbenzene (SDB) copolymer, the reactant CH3OH and the product MMA during the adsorption of H2O on a Pd surface were investigated. For a Pd/SDB catalyst, the interactions between the active component and the carrier were first calculated using Pd4 clusters. The results implied that Pd4 clusters were chemisorbed on the SDB carrier. By comparing the adsorption energy of H2O molecules on Pd4 clusters with or without SDB, it was found that the adsorption energy of the former was reduced by about 50%, indicating that the hydrophobic carrier SDB reduced the adsorption of H2O on Pd4 clusters. This was also confirmed by the results for the partial density of states, differences in charge density and comparative Mulliken charge analysis. The influences of the reactant CH3OH and the product MMA on the adsorption of H2O were investigated using the Pd(111) surface. The results of co-adsorption simulations showed that some of the electrons on CH3OH molecules were transferred to H2O molecules that strengthened the electronic interaction between H2O molecules and the Pd surface and led to a change in the adsorption of isolated H2O molecules from physisorption to chemisorption. However, the product MMA when chemisorbed on the Pd surface had little effect on the adsorption of H2O molecules on the Pd(111) surface.


 Please wait while we load your content...
Please wait while we load your content...