Mechanism of protein–RNA recognition: analysis based on the statistical mechanics of hydration†
Abstract
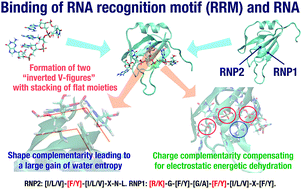
We investigate the RBD1–r(GUAGU) binding as a case study using all-atom models for the biomolecules, molecular models for water, and the currently most reliable statistical–mechanical method. RBD1 is one of the RNA-binding domains of mammalian Musashi1 (Msi1), and r(GUAGU) contains the minimum recognition sequence for Msi1, r(GUAG). We show that the binding is driven by a large gain of configurational entropy of water in the entire system. It is larger than the sum of conformational-entropy losses for RBD1 and r(GUAGU). The decrease in RBD1–r(GUAGU) interaction energy upon binding is largely cancelled out by the increase in the sum of RBD1–water, r(GUAGU)–water, and water–water interaction energies. We refer to this increase as “energetic dehydration”. The decrease is larger than the increase for the van der Waals component, whereas the opposite is true for the electrostatic component. We give a novel reason for the empirically known fact that protein residues possessing side chains with positive charges and with flat moieties frequently appear within protein–RNA binding interfaces. A physical picture of the general protein–RNA binding mechanism is then presented. To achieve a sufficiently large water-entropy gain, shape complementarity at the atomic level needs to be constructed by utilizing the stacking and sandwiching of flat moieties (aromatic rings of the protein and nucleobases of RNA) as fundamental motifs. To compensate for electrostatic energetic dehydration, charge complementarity becomes crucial within the binding interface. We argue the reason why the RNA recognition motif (RRM) is the most ubiquitous RNA binding domain.


 Please wait while we load your content...
Please wait while we load your content...