Penetrating probability and cross section of the Li+–C60 encapsulation process through an ab initio molecular dynamics investigation
Abstract
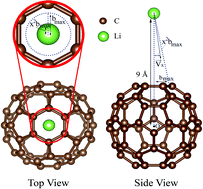
The endohedral complex system of Li+–C60 has been shown to possess interesting applications in photovoltaics, supramolecular chemistry, and functionalized materials. In this study, we perform a theoretical investigation of Li+ encapsulation within a C60 cage by employing an ab initio molecular dynamics approach. The Li+ cation is positioned 9 Å away from the C60 center of mass, and fired towards a randomized spot in a six-membered ring with a certain level of inletting energy, which is 7.5 eV, 9 eV, 12 eV, or 15 eV. In total, 2000 samples of MD trajectories are investigated. Our statistical results yielded a penetrating probability in the range of 0.8% to 15.6% with respect to the above inletting energy, while the cross section ranges from 0.006 Å2 to 0.123 Å2. Moreover, we observed that the penetrating probability exhibited direct proportionality to the inletting energy. Hence, we can determine that the minimum required inletting energy for reaction occurrence is 6.6 eV. Overall, it seems difficult for Li+ to penetrate through the sp2-carbon wall, because a very high inletting energy is required to open the entrance. At the same time, Li+ must approach closely to the center of a six-membered ring to enhance the penetration probability.


 Please wait while we load your content...
Please wait while we load your content...