Conformational analysis of a supramolecular synthon: a case study of 8-hydroxyquinoline†
Abstract
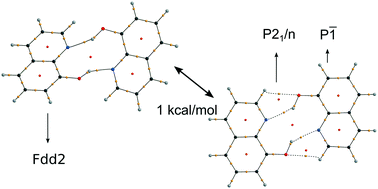
A detailed comparative study of three polymorphic forms of 8-hydroxyquinoline (8HQ) was performed, by using X-ray diffraction, quantum chemical methods and different approaches to crystal packing analysis. The supramolecular synthon in 8HQ and its derivatives is a H-bonded dimer, which can exist in either a twisted or a co-planar conformation. According to B3LYP-D3/def2-TZVPP calculations, the twisted conformation with C2 symmetry corresponds to the energy minimum, whereas the planar C2h geometry is a transition state with an imaginary mode of 19.4i cm−1. Despite significant differences in geometry, the energy difference between the two conformations is only 1.0 kcal mol−1, since the unfavorable out-of-plane rotation of the OH group in the twisted conformation is almost compensated by the stronger H-bond. The relative stability of 8HQ polymorphs is therefore defined not by the strongest interactions in the synthon but by other intermolecular interactions. The results demonstrate that conformational analysis of supramolecular synthons is important in understanding the differences and similarities of crystal structures. Furthermore, the term “conformational polymorphism” can be used in a broader sense, if conformations of a supramolecular synthon are considered in the same way as molecular conformations, at least for strong intermolecular interactions such as H-bonds.


 Please wait while we load your content...
Please wait while we load your content...