Photo- and electro-luminescence of three TADF binuclear Cu(i) complexes with functional tetraimine ligands†
Abstract
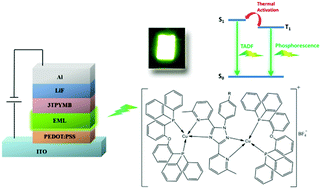
Three new binuclear cuprous complexes with similar tetraimine ligands [Cu2(pytzph)(POP)2](BF4)2 (1), [Cu2(pytzphcf)(POP)2](BF4)2 (2) and [Cu2(pytzphcz)(POP)2](BF4)2 (3), (pytzph = 6,6′-(1-phenyl-1,2,4-triazole-3,5-diyl)bis(2-methylpyridine), pytzphcf = 6,6′-(1-(4-(trifluoromethyl)phenyl)-1,2,4-triazole-3,5-diyl)bis(2-methylpyridine), pytzphcz = 9-(4-(3,5-bis(6-methylpyridin-2-yl)-1,2,4-triazol-1-yl)phenyl)-carbazole and POP = bis[2-(diphenylphosphine)phenyl]ether), have been synthesized and characterized in order to compare the different effects of substituent groups on the photoluminescence (PL) and electroluminescence (EL) properties. These complexes exhibit highly efficient green thermally activated delayed fluorescence (TADF) with short decay times (5.5–16 μs) and high photoluminescence quantum yields (up to 79%) at room temperature in the solid form. These complexes have essentially identical emission energy. However, the influence of the substituents on the photoluminescence and electroluminescence efficiencies is evident. Complex 3 with the carbazole group shows the highest efficiency in terms of both PL and EL, exhibiting an EQE of 8.3%, a CE of 27.1 cd A−1 and a peak brightness of 2525 cd cm−2 in the solution-processed OLED, while complex 2 with a trifluoromethyl appendage exhibits poorer quantum efficiency than the others.


 Please wait while we load your content...
Please wait while we load your content...