Ladder-type oligo(p-phenylene)s with D–π–A architectures: design, synthesis, optical gain properties, and stabilized amplified spontaneous emission†
Abstract
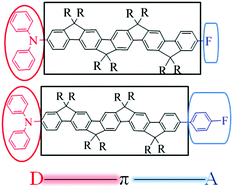
A novel family of rigid ladder-type oligo(p-phenylene)s with donor–π–acceptor (D–π–A) architectures (n)L-F/(n)L-Ph-F (n = 2–4) end-capped with diphenylamino and fluorophenyl/fluorine units have been designed, synthesized and explored as gain media for organic lasers. The resulting materials demonstrated excellent thermal stability with a high degradation temperature (Td) over 400 °C. The extension of the π-conjugated bridge length between the donor and acceptor units successfully depressed the crystallization tendency of oligo(p-phenylene)s, resulting in enhanced glassy temperature (Tg) and improved morphological stability in neat films. The amplified spontaneous emission (ASE) threshold (Eth) decreases with an extension in the conjugation length of the oligo(p-phenylene)s. In particular, for 4L-Ph-F with the longest conjugation length, the ASE threshold is determined to be as low as 1.97 μJ cm−2 with a high net gain coefficient over 90 cm−1 and a rather low loss coefficient of α = 2.0 cm−1. One dimensional distributed feedback (1D DFB) lasers demonstrated lasing thresholds of 5.3 nJ pulse−1 (0.44 kW cm−2, 2.2 μJ cm−2) and 1.3 nJ pulse−1 (0.1 kW cm−2, 0.5 μJ cm−2) for 4L-F (at 460 nm) and 4L-Ph-F lasers (at 471 nm), respectively. It should be noted that the Eth value of all the ladder-type samples (n)L-Ph-F (n = 2–4) remains almost the same upon increasing the annealing temperature even up to 220 °C. The high gain and low loss with excellent thermal and optical stability have rendered these rigid D–π–A ladder-type materials advantageous as robust gain media for organic lasers.
- This article is part of the themed collection: Journal of Materials Chemistry C Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...