Dendritic nanotubes self-assembled from stiff polysaccharides as drug and probe carriers†
Abstract
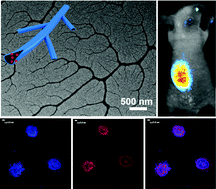
Dendritic nanotubes (DNTs) with hydrophobic cavities were constructed directly from rigid branched β-1,3-D-glucan (AF1) in aqueous solution, and the AF1 sample was isolated from the fruiting bodies of Auricularia auricula-judae, a household nutritional food. The structure of AF1 dendritic nanotubes was demonstrated with a transmission electron microscope (TEM) and a scanning electron microscope (SEM), and a schematic diagram was proposed to describe the formation process, which was supported by the results of static/dynamic light scattering (SLS/DLS) and atomic force microscopy (AFM). In solution, a sequential self-assembly of the AF1 chains in a parallel manner occurred to form lamellas followed by self-curling into nanotubes with the mean diameters from 20 to 80 nm, depending on the concentration and molecular weight of AF1, through hydrogen bonding and hydrophilic/hydrophobic interaction. As a result of the dendritic structure, the AF1 aggregates exhibited highly condensed hydrophobic regions, which could be used as carriers to achieve a high concentration of the target molecules. In our findings, the anticancer drug DOX and the fluorescent probe TPA-BMO could be loaded into the hydrophobic region of DNTs. Interestingly, DOX-loaded DNTs of AF1 exhibited high drug loading capacity and pH-triggered sustained release behaviors (>23 days) with reduced cytotoxicity in vitro. Moreover, the bioimaging experiment demonstrated that TPA-BMO-loaded DNTs of AF1 induced stronger fluorescence intensity than TPA-BMO alone, and maintained a longer duration time (18 days) in vivo. Therefore, the DNTs of AF1 have promising applications as bioactive carriers, especially in the fields of drug delivery and bioimaging.


 Please wait while we load your content...
Please wait while we load your content...