Mediating physicochemical properties and paclitaxel release of pH-responsive H-type multiblock copolymer self-assembly nanomicelles through epoxidation†
Abstract
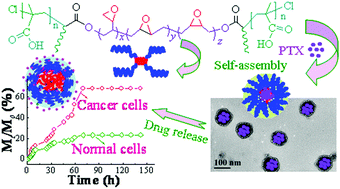
pH-Sensitive H-type multiblock copolymers, namely, poly(methacrylic acid)2-block-epoxidized hydroxyl-terminated polybutadiene-block-poly(methacrylic acid)2 (PMAA2-b-epoHTPB-b-PMAA2), were synthesized by atom-transfer radical polymerization and subsequent in situ epoxidation by peracetic acid and characterized by 1H NMR, FT-IR and SEC techniques. The impact of epoxidation on the physicochemical and biomedical properties of copolymer self-assembly micelle nanoparticles was investigated by fluorescence spectrometry, DLS, TEM and an MTT assay. The experimental results indicated that epoxidation resulted in the formation of more stable copolymer micelle nanoparticles with a lower critical micelle concentration, smaller micelle size, and higher loading capacity and encapsulation efficiency of drugs than those without epoxidation. In particular, epoxidized copolymer micelle nanoparticles exhibited reasonable pH sensitivity at a pH of 5.3–5.6. The hydrophobic anticancer drug paclitaxel (PTX) displayed faster release rates from epoxidized nanomicelles than from unepoxidized nanomicelles in a PBS solution of a pH of 4.8–6.6, whereas in PBS of a pH of 7.4 smaller amounts of PTX were released from epoxidized nanomicelles than from unepoxidized nanomicelles. Epoxidized copolymer nanomicelles were reasonably biodegradable after the drug was released, and their degradation rate was faster than that of their unepoxidized counterparts. An MTT assay was performed to determine the biocompatibility of epoxidized copolymer micelle nanoparticles and the anticancer activities of PTX-loaded nanomicelles, which were important for applications in the therapy of cancers as a controlled-release drug carrier.


 Please wait while we load your content...
Please wait while we load your content...