Aromatic primary monoamine-based fast-response and highly specific fluorescent probes for imaging the biological signaling molecule nitric oxide in living cells and organisms†
Abstract
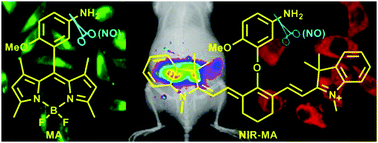
Nitric oxide (NO) is an important cellular signaling molecule involved in many physiological and pathological processes. To probe its spatiotemporal information in biosystems, a large number of NO fluorescent probes have been exploited in the past ten years. Among them, the o-phenylenediamine-based probes are the earliest developed and most versatile NO fluorescent probes to date. However, there are still limitations such as relatively long response time, possible interference by dehydroascorbic acid (DHA)/methylglyoxal (MGO), and pH-sensitivity of their fluorescence signals. In this work, we present two aromatic primary monoamine-based NO fluorescent probes, MA and NIR-MA, and explore the reductive deamination reaction of the electron-rich p-methoxyaniline group with NO under aerobic conditions. The superiority of both probes is illustrated by their quick, stable, sensitive, and specific fluorescence off–on responses for NO over a series of biologically relevant interfering species, including reactive oxygen species, DHA/MGO, biothiols, and metal ions. Coupled with good cell permeability and low cytotoxicity, the two probes have successfully been applied to imaging the endogenous NO in RAW 264.7 macrophages stimulated by LPS/IFN-γ. Moreover, the fluorescence response of NIR-MA for NO occurs in the physiologically favorable NIR region, enabling its further use to image endogenous NO in an inflamed mouse model.


 Please wait while we load your content...
Please wait while we load your content...