General promoting effect of polydopamine on supported noble metal catalysts†
Abstract
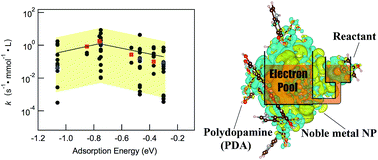
Metal-support interaction and corresponding interface regulation are of great importance for the preparation of well-defined, supported metal catalysts with outstanding performance. Herein a joint experimental-theoretical study has been conducted to investigate the role of polydopamine (PDA) as an interface facilitator for supported noble metal catalysts. The effects of PDA between various noble metals (Pt, Pd, Au, Ag) and a support (SBA-15) have been systematically investigated in the model reaction of 4-nitrophenol (4-NP) reduction by NaBH4. Based on the kinetic model of 4-NP reduction by NaBH4 and a set of density functional theory calculations on different 4-NP binding energies, a volcano-shaped relationship plot is obtained. The volcano-shaped dependence of the reaction rate constant on 4-NP adsorption energy is observed for supported noble metal catalysts with/without PDA. The general relationship indicates that PDA can effectively improve catalytic performance via adjusting 4-NP adsorption energy to a more suitable value. The results of charge density difference and Bader charge analysis illustrate the establishment of an ‘electron pool’ by PDA in the system to achieve controllable electronic charge redistribution for an optimized binding when the reactant molecule adsorbs on the metal nanoparticle.


 Please wait while we load your content...
Please wait while we load your content...