Biscoumarin-containing acenes as stable organic semiconductors for photocatalytic oxygen reduction to hydrogen peroxide†
Abstract
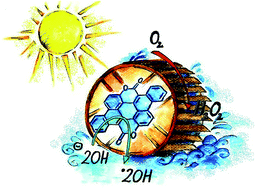
Conversion of solar energy into chemical energy in the form of hydrogen peroxide and other reactive oxygen species has been predicted to be an efficient strategy, yet few organic materials systems support these types of photochemical conversion reactions. Herein we report a simple synthetic route to yield biscoumarin-containing acenes, semiconducting small molecules with exceptional stability and tunable electrochemical and electrical properties. We find that these semiconductors are photo(electro)catalysts capable of reducing oxygen to hydrogen peroxide. Visible light irradiation of thin films on insulating substrates in pure water results in H2O2 photogeneration with water as the sacrificial electron donor. Thin films on conducting substrates are robust catalytic photocathodes for producing H2O2. These semiconductor photoelectrodes retain their catalytic properties in a pH range from 2–13. Photocatalytic or photoelectrocatalytic deployment of biscoumarin-containing acenes does not lead to measurable degradation. This work demonstrates a strategy to synthesize stable organic semiconductors not only suitable for thin-film electronic devices but also next-generation photocatalytic concepts.


 Please wait while we load your content...
Please wait while we load your content...