Retracted Article: Novel mesoporous amorphous B–N–O–H nanofoam as an electrode for capacitive dye removal from water†
Abstract
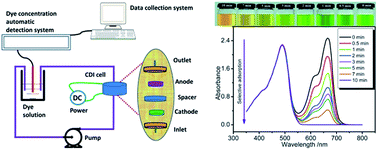
Novel mesoporous B–N–O–H nanofoam was prepared using CuB23 and ammonium chloride precursors under O2 in ionic liquids via a solution plasma technique. It had a large specific surface area of 1023 m2 g−1, and exhibited high electrical conductivity of 20.8 S m−1 and good specific capacity of 184 F g−1 in 600 mg L−1 methylene blue aqueous solution at a current density of 0.2 mA cm−2. Furthermore, the nanofoam exhibited a high adsorption capacity for cationic dyes, especially methylene blue, with well-controlled recycling behavior. Meanwhile, the methylene blue/acid orange 7 mixture could be easily separated for reuse by the strong electrostatic interaction. The good separation of methylene blue/Rhodamine B was attributed to the stronger coordination between methylene blue and the B–N–O–H nanofoams in comparison with that of Rhodamine B, and the advantage of the smaller methylene blue in competitive adsorption at higher flow rates. These results indicate that the present B–N–O–H nanofoam may have promising applications in the field of water purification, including enrichment or even total separation of dyes during wastewater treatment for reuse.


 Please wait while we load your content...
Please wait while we load your content...