Optimizing the ORR activity of Pd based nanocatalysts by tuning their strain and particle size†
Abstract
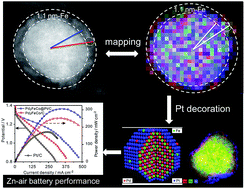
Controlling of the particle size and surface strain is the key to tuning the surface chemistry and optimizing the catalytic performance of electrocatalysts. Here, we show that by introducing both Fe and Co into Pd lattices, the surface strain of Pd nanocatalysts can be tuned to optimize their oxygen reduction activity in both fuel cells and Zn–air batteries. The Pd2FeCo/C alloy particles are uniquely coated with an ultrathin Fe2O3 shell which is in situ formed during a thermal annealing treatment. The thin shell acts as an effective barrier that prevents the coalescence and ripening of Pd2FeCo/C nanoparticles. Compared with Pd/C, Pd2FeCo/C exhibits higher catalytic activity and long-term stability for the ORR, signifying changes in catalytic behavior due to particle sizes and strain effects. Moreover, by spontaneous decoration of Pt on the surface of Pd2FeCo/C, the Pd2FeCo@Pt/C core@shell structure was formed and the Pt mass activity was about 37.6 and 112.5 times higher than that on Pt/C in a 0.1 M HClO4 and KOH solution at 0.9 V, respectively, suggesting an enhanced ORR performance after Pt decoration. More interestingly, Pd2FeCo@Pt/C also shows a power density of ∼308 mW cm−2, which is much higher than that of Pt/C (175 mW cm−2), and excellent durability in a home-made Zn–air battery.


 Please wait while we load your content...
Please wait while we load your content...