Hydrogen generation via hydrolysis of magnesium with seawater using Mo, MoO2, MoO3 and MoS2 as catalysts
Abstract
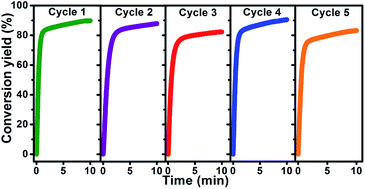
Hydrogen generation is one of the enabling technologies for realization of hydrogen economy. In this study, we developed a high-performance hydrogen generation system using the transition metal Mo and its compounds (MoS2, MoO2, and MoO3) for catalyzing the hydrolysis of Mg composites in seawater. These Mg-based composites for the hydrolysis process were synthesized through a simple planetary ball mill technique. The results demonstrate that small amounts of added MoS2 could significantly accelerate and enhance the hydrolysis reaction of Mg in seawater. In particular, the Mg–10 wt% MoS2 composite releases 838 mL g−1 hydrogen in 10 min (about 89.8% of the theoretical hydrogen generation yield), and the recycled catalysts exhibit high cycle stability, which is the most significant achievement in this study. In addition, Mo, MoO2, and MoO3 also showed similar enhancement in the hydrolysis reaction of Mg. The activation energies for the hydrolysis of Mg decreased from 63.9 kJ mol−1 to 27.6 kJ mol−1, 20.4 kJ mol−1, 14.3 kJ mol−1, and 12.1 kJ mol−1 on introducing Mo, MoS2, MoO2, and MoO3, respectively. The attractive hydrolysis performance of the composites of Mg milled with Mo and its compounds in seawater may shed light on future developments of hydrogen generation technologies.


 Please wait while we load your content...
Please wait while we load your content...