Simultaneous enhancement of the molecular planarity and the solubility of non-fullerene acceptors: effect of aliphatic side-chain substitution on the photovoltaic performance†
Abstract
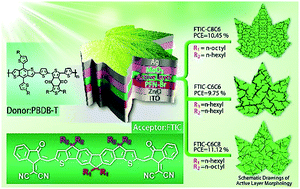
Three planar nonfullerene acceptors (FTIC-C8C6, FTIC-C6C6 and FTIC-C6C8) comprising a central fluorenedicyclopentathiophene (FT) core and two 2-methylene-(3-(1,1-dicyanomethylene)-indanone) terminal groups are designed and synthesized. The coplanarity of the molecular backbone can be maintained through a locked conformation via intramolecular noncovalent interactions. The solubility of these nonfullerene acceptors is very good because the FT core can bear enough flexible aliphatic side-chain substitutions. Thus, the dilemma of the planarity–solubility tradeoff can be minimized. Through changing the length of the six flexible aliphatic side chains at the central FT core, we can easily adjust the π–π interactions of nonfullerene acceptors and optimize the nanoscale morphology of the photoactive layers. Among these three small molecular acceptors, FTIC-C6C8 based active layers show the best morphology together with the highest electron and hole mobility. These inherent advantages of FTIC-C6C8 guarantee it a high power conversion efficiency of 11.12% when used in non-fullerene polymer solar cells with a wide-bandgap polymer donor PBDB-T. Our results provide an appropriate molecular design strategy for building high-performance nonfullerene acceptors and show that optimizing alkyl-side chains is a very effective way to further improve the photovoltaic performance of devices.


 Please wait while we load your content...
Please wait while we load your content...