Graphene-coated copper nanowire networks as a highly stable transparent electrode in harsh environments toward efficient electrocatalytic hydrogen evolution reactions†
Abstract
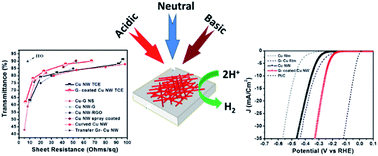
Copper nanowire networks (NWs) coated with a graphene layer through a carbon-enclosed chemical vapor deposition technique at a low temperature of 400 °C with a low sheet resistance of 23.2 Ω sq−1 and a high transmittance of 83.4%, which is comparable to the typical values of tin-doped indium oxide (ITO), as a transparent conducting electrode were demonstrated. The graphene-coated copper NW networks retain a low sheet resistance of less than 25 Ω sq−1 even after annealing at a temperature of 240 °C in a pure oxygen environment for 1 h, while a sheet resistance less than 100 Ω sq−1 can still be maintained in natural sea water, and acidic and basic solutions. Their highly stable features in harsh environments make these graphene-coated copper nanowire networks suitable as a catalyst toward high-efficiency hydrogen evolution reactions (HERs) with a low overpotential of 252 mV at 10 mA Cm−2 and a low Tafel slope of 67 mV dec−1. The non-corrosive and anti-oxidant graphene-coated copper nanowire networks could be used as an alternative transparent conducting electrode in harsh environments, such as in tandem photocatalytic water splitting.


 Please wait while we load your content...
Please wait while we load your content...