Elucidating the diffusion pathway of protons in ammonium polyphosphate: a potential electrolyte for intermediate temperature fuel cells†
Abstract
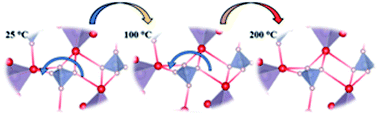
Ammonium polyphosphate (NH4PO3) is a potential electrolyte material for intermediate temperature fuel cells (150–250 °C). The crystal structure of NH4PO3, including the H positions, is unravelled by neutron powder diffraction (NPD) data by successive Fourier synthesis from the polyphosphate backbone. The structure consists of zig–zag chains aligned along the [001] directions of tetrahedral phosphate PO4 units that are connected through O3 atoms with a P–O3–P angle of 126.3(5)°. The proton conductivity mechanism of NH4PO3 is clarified from the thermal evolution of the structure. It shows that some H atoms subtly shift at high temperatures, resulting in a weakening of certain H-bonds, thus increasing the lability of those H atoms involved in the proton conduction mechanism. Conductivity measurements in humid air and H2 of NH4PO3 show high proton conductivities of 1.2 × 10−5 to 2.61 × 10−3 S cm−1 and 2.2 × 10−5 to 2.69 × 10−3 S cm−1, respectively, in the temperature range of 50 °C to 275 °C.


 Please wait while we load your content...
Please wait while we load your content...