Heteropolyacids embedded in a lipid bilayer covalently bonded to graphene oxide for the facile one-pot conversion of glycerol to lactic acid†
Abstract
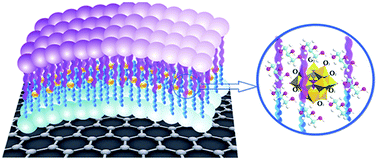
A new two-step strategy for the functionalization of graphene oxide (GO) with heteropolyacids (HPAs) and the catalytic activity of the so-obtained materials in the conversion of glycerol to lactic acid (LA) are reported. Covalent bonding of a a-NH–(CH2)2–NH2+–CH2(CH2)8CH3 lipid-like bilayer to the GO surface is followed by embedding of HPAs to give HPMo@lipid(n)/GO hybrid materials, where n represents the length of the diamine carbon chain (n = 2, 4, 6, 8, and 10). The HPAs are tightly surrounded by the lipid bilayer through electrostatic interactions with the protonated amine groups and are highly resistant to environmental changes and leaching from the GO. The hydrophilicity of HPAs and GO and the hydrophobic properties of the lipid bilayer can be easily controlled by changing the length of the alkyl chain, and the redox potential varies as the distance between HPAs and GO varies. The materials show excellent catalytic activity in glycerol cascade conversion to LA, with HPMo@lipid(4)/GO achieving the highest reported efficiency to date, with 90% yield at 97% conversion under mild conditions (1 M glycerol, 60 °C, 3.5 h, and 10 bar O2). Such high efficiency is attributed to the combination of suitable redox potentials, balanced hydrophilic and hydrophobic properties, and the capillary-like reactor formed by the wall of the lipid bilayer, which enhances the adsorption of O2 and glycerol around catalytic active sites. HPMo@lipid(4)/GO functions in the absence of an organic solvent and base, with a high concentration of glycerol (9.2 wt%) and in the presence of methanol and other impurities from biodiesel production, with crude glycerol giving almost 87% yield. No structural changes or leaching of HPAs from GO occurs during the reaction or with recycling up to fifteen times.


 Please wait while we load your content...
Please wait while we load your content...