Multicomponent (Mo, Ni) metal sulfide and selenide microspheres with empty nanovoids as anode materials for Na-ion batteries†
Abstract
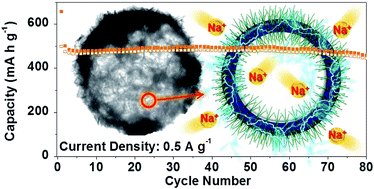
Unique-structured MoS2–Ni9S8–C, MoSe2–NiSe–C, MoS2–Ni9S8, and MoSe2–NiSe–NiSe2 composite microspheres were prepared by pilot-scale spray drying and a subsequent post-treatment process. Dextrin, used as the carbon source, played a key role in the formation of unique-structured multicomponent metal sulfide and selenide microspheres with and without carbon. The empty nanovoids formed by decomposition (or carbonization) of phase-separated dextrin during the spray drying process were uniformly distributed within the multicomponent metal sulfide and selenide composite microspheres. The discharge capacities of MoS2–Ni9S8–C, MoSe2–NiSe–C, MoS2–Ni9S8, and MoSe2–NiSe–NiSe2 for the 80th cycle at a current density of 0.5 A g−1 for sodium-ion storage were 366, 386, 459, and 291 mA h g−1, respectively, and the respective capacity retentions measured from the second cycle were 91, 102, 92, and 64%. The carbon-free MoS2–Ni9S8 microspheres exhibited excellent rate performance and their discharge capacities decreased slightly from 559 to 428 mA h g−1 as the current densities increased from 0.1 to 3 A g−1. The MoS2–Ni9S8–C composite microspheres, with high structural stability during repeated sodium-ion insertion and deinsertion, showed extremely long-term cycling performance for 1000 cycles.


 Please wait while we load your content...
Please wait while we load your content...