Two-dimensional CoNi nanoparticles@S,N-doped carbon composites derived from S,N-containing Co/Ni MOFs for high performance supercapacitors†
Abstract
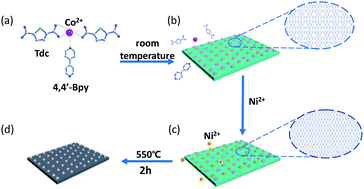
Due to their controllable morphologies, tunable porous structures, diverse compositions and easy fabrication, metal–organic frameworks (MOFs) are an ideal class of precursor material to develop high performance carbon-based materials for energy applications. In this work, two-dimensional (2D) Co/Ni MOFs nanosheets with a molar ratio of Co2+ to Ni2+ of 1 : 1 were first synthesized at room temperature using thiophene-2,5-dicarboxylate (Tdc) and 4,4′-bipyridine (4,4′-Bpy) as organic linkers. As a precursor material, the as-synthesized 2D Co/Ni MOFs nanosheets were further pyrolized at 550 °C in N2 atmosphere to incorporate 2D CoNi alloy nanoparticles into S, N-doped carbon nanosheets (CoNi@SNC) with a surface area of 224 m2 g−1, a porous structure, and good conductivity. Interestingly, it was found that the 2D Co/Ni MOFs nanosheets can be directly used as electrode materials for supercapacitors, delivering a specific capacitance of 312 F g−1 at 1 A g−1, whereas CoNi@SNC derived from its MOFs precursor as an electrode material for supercapacitors exhibits a much higher specific capacitance (1970, 1897 and 1730 F g−1 at 1, 2, 5 A g−1, respectively) with long cycling life (retaining 95.1% of the value at 10 A g−1 after 3000 cycles) and excellent rate capability at a high charge/discharge current. Further, an asymmetric supercapacitor device was constructed with CoNi@SNC as the positive electrode and active carbon as the negative electrode, exhibiting an energy density of 55.7 W h kg−1 at a power density of 0.8 kW kg−1 with lifetime stability up to 4000 charge–discharge cycles (capacitance retention of ∼90.6%). The results demonstrate that electrochemical activation-generated CoNi oxides/oxyhydroxides on the surface of the CoNi alloy nanoparticles in alkaline electrolyte during electrochemical measurements are the electrochemical active species of the CoNi@SNC-constructed supercapacitor. Additionally, the high performance of the CoNi@SNC-constructed supercapacitor can be collectively attributed to its relatively high surface area, which is favourable for the exposure of electrochemical active sites; its porous structure, which promotes redox-related mass transport; and the combination of CoNi alloy nanoparticles with graphitic carbon, which functions as an electron collector to improve electron transfer.


 Please wait while we load your content...
Please wait while we load your content...