Fine-crystallized LDHs prepared with SiO2 spheres as highly active solid base catalysts†
Abstract
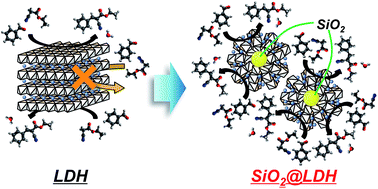
Fine-crystallized layered double hydroxides are prepared via the co-precipitation method with the coexistence of SiO2 spheres (SiO2@LDH), and their base catalysis and structural properties are investigated. The as-prepared SiO2@LDHs exhibit higher base catalysis for the Knoevenagel condensation than conventional LDHs prepared via the co-precipitation method in the absence of SiO2 spheres. Such an increase in the activity for base catalysis is also observed in various types of SiO2@LDHs with different metal compositions (M2+: Mg2+ or Ni2+, M3+: Al3+ or Ga3+, and M2+/M3+: 1 or 3). X-ray diffraction (XRD) measurement suggests that the addition of SiO2 induces decreases in the LDH crystallite size. The results of transmission electron microscopy – energy dispersive X-ray spectroscopy (TEM-EDS) and 29Si cross polarization magic angle spinning nuclear magnetic resonance (29Si CP-MAS NMR) on SiO2@Mg–Al LDH suggest that the crystals of Mg–Al LDH are immobilized on the SiO2 surface through Si–O–Al and Si–O–Mg covalent bonds. According to these results, we conclude that the co-precipitation method in the presence of colloidal spherical SiO2 seeds, particularly possessing a 40 nm diameter, result in highly-dispersed points of LDH crystal growth on the SiO2 surface, which lead to the generation of fine-crystallized highly active LDH nanocrystals.


 Please wait while we load your content...
Please wait while we load your content...